The Benefit-Risk Evaluation Challenge
Pharmaceutical teams face mounting pressure to demonstrate drug safety and efficacy across every stage, from regulatory submission to market access and post approval surveillance. Yet traditional evaluation methods create critical vulnerabilities.
Time-Consuming Manual Analysis
Full benefit-risk assessments across the drug lifecycle can span 12-18 months, while individual PSUR/PBRER cycles alone take 12-16 weeks. Each cycle is a manual, resource-intensive process that delays critical go/no-go decisions and market authorization filings.
Incomplete Evidence Synthesis
Fragmented data sources and manual literature reviews miss critical safety signals, leaving gaps in regulatory submissions and HTA dossiers.
High Failure
Costs
With 9 out of 10 drugs failing (57% efficacy issues, 17% safety concerns) at an average cost of $2.3B per approval (Source: Tufts CSDD, 2020), incomplete benefit-risk intelligence compounds financial risk across the portfolio.
One Platform. Complete Visibility.
Six stages. From clinical framing to automated outputs
100B+ data points | 24 AI models | Trusted by top 10 pharma companies
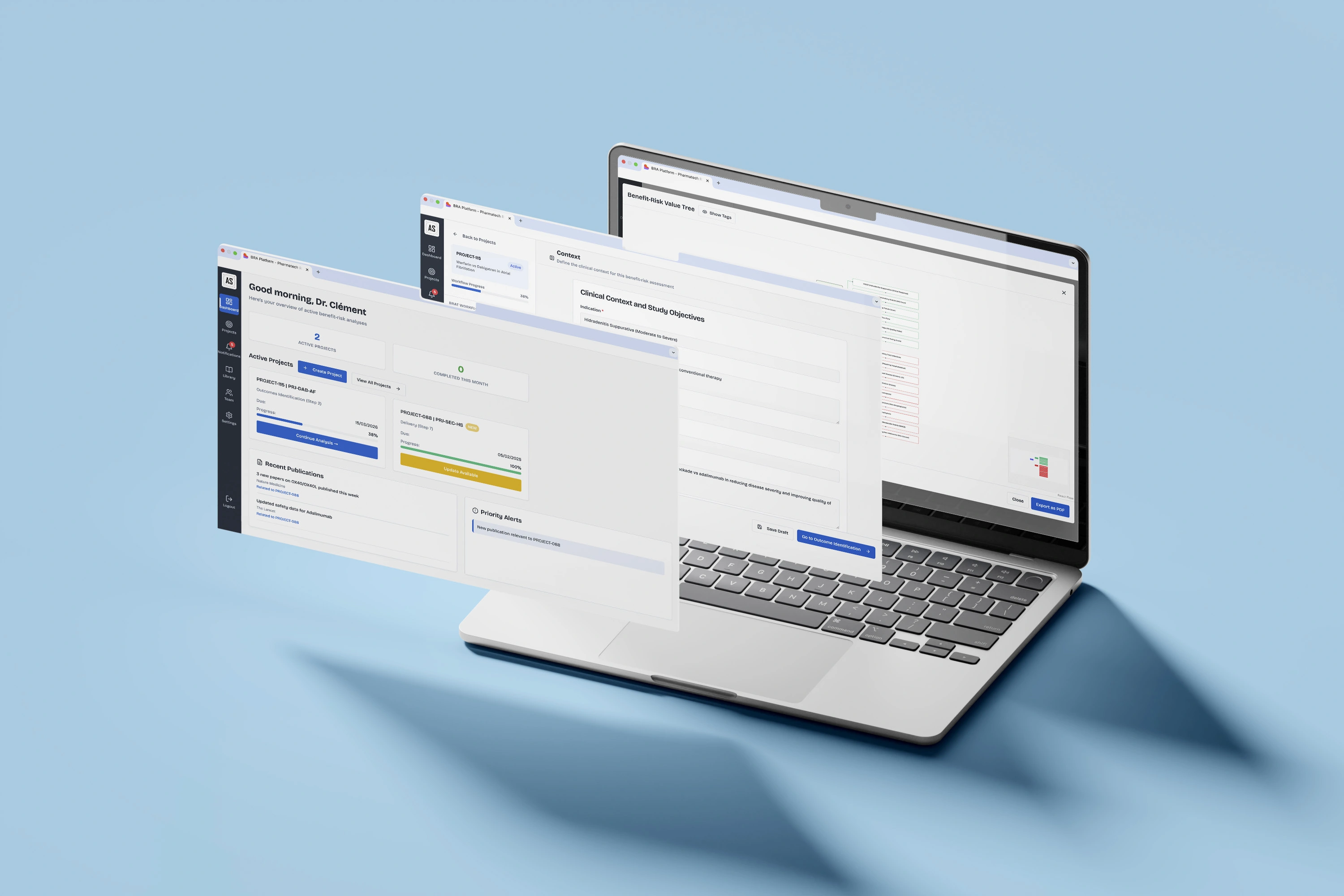
Clinical Framing
Set protocol parameters (drug, dosage, comparator), establish key endpoints and decision criteria, and frame your regulatory strategy: ensuring alignment across clinical, regulatory, and market access teams.
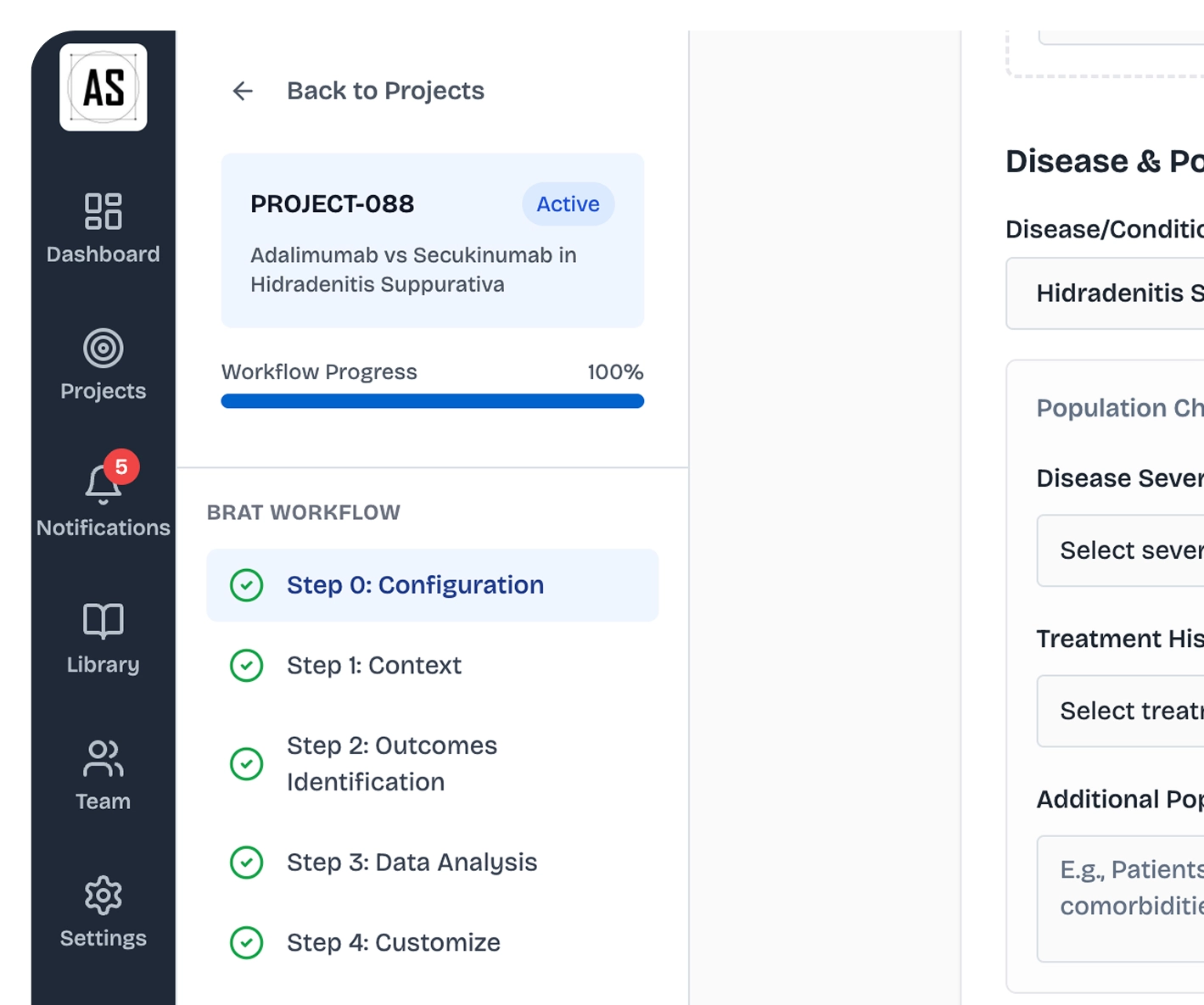

Indication in. Outcome list out
ArcaScience pre-populates your benefit and risk outcomes from your drug, comparator, and indication. You validate. Not search.
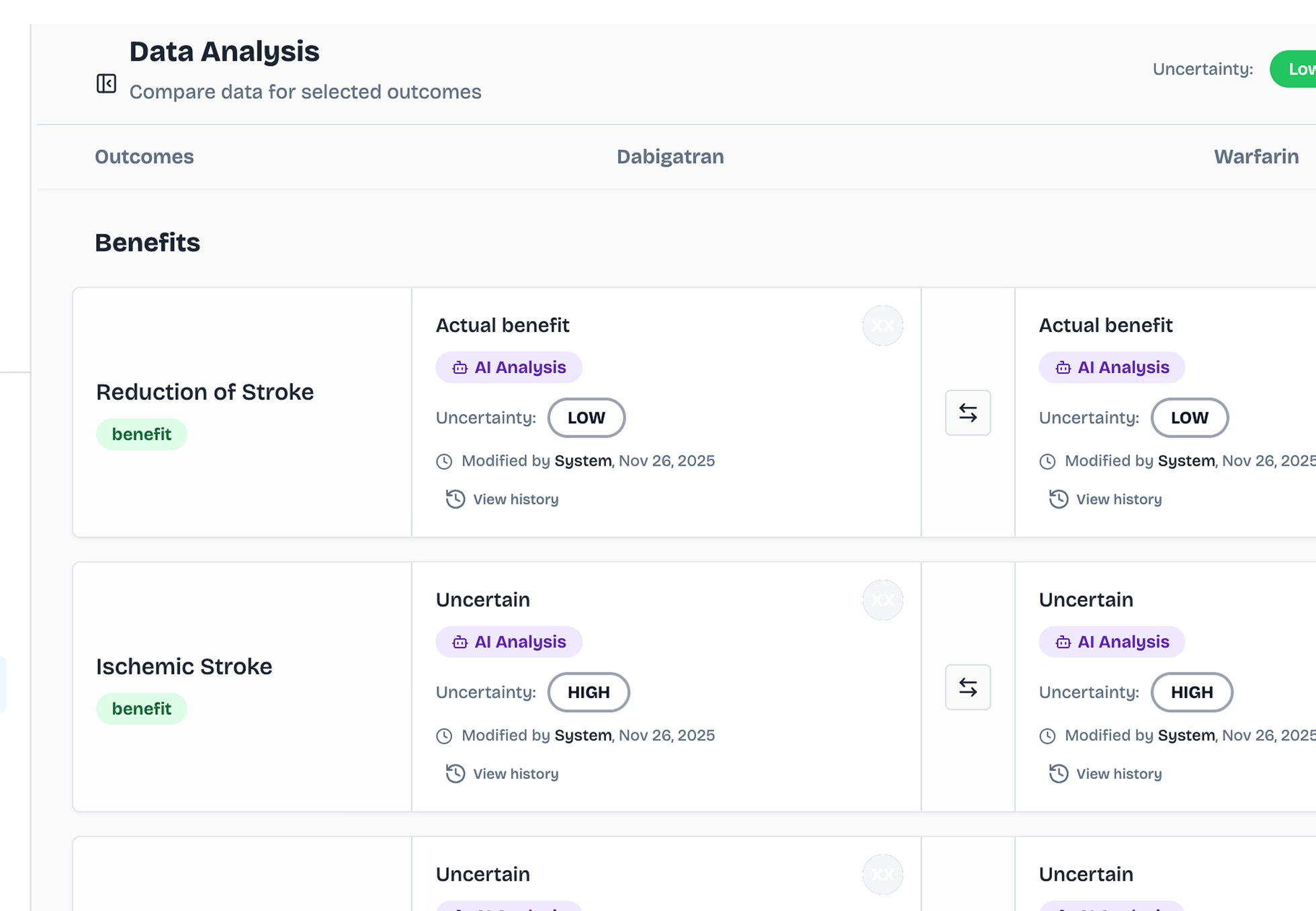
Every outcome assessed
ArcaScience runs the comparative assessment for each selected outcome, benefits and risks, drug and comparator, side by side. AI-generated analysis with evidence scoring. No manual data extraction, no spreadsheet.
Your judgment, built into the model
Remove irrelevant outcomes, reorder benefits and risks, and adjust weightings to reflect your submission strategy. The Value Tree updates in real time.
Your assessment, ready to defend
No more assembling results from scattered sources. ArcaScience structures your benefit–risk outputs in a single, interpretable view, ready to present to regulators, HTA bodies, or internal stakeholders.
"For the first time in our history, we successfully achieved a benefit-risk drill-down prediction based on 9x more insights, anticipating thromboembolic risks and re-routing millions in development funds."
Global R&D Team
Philippe Peyre, Secretary General, Vice President, Sanofi.
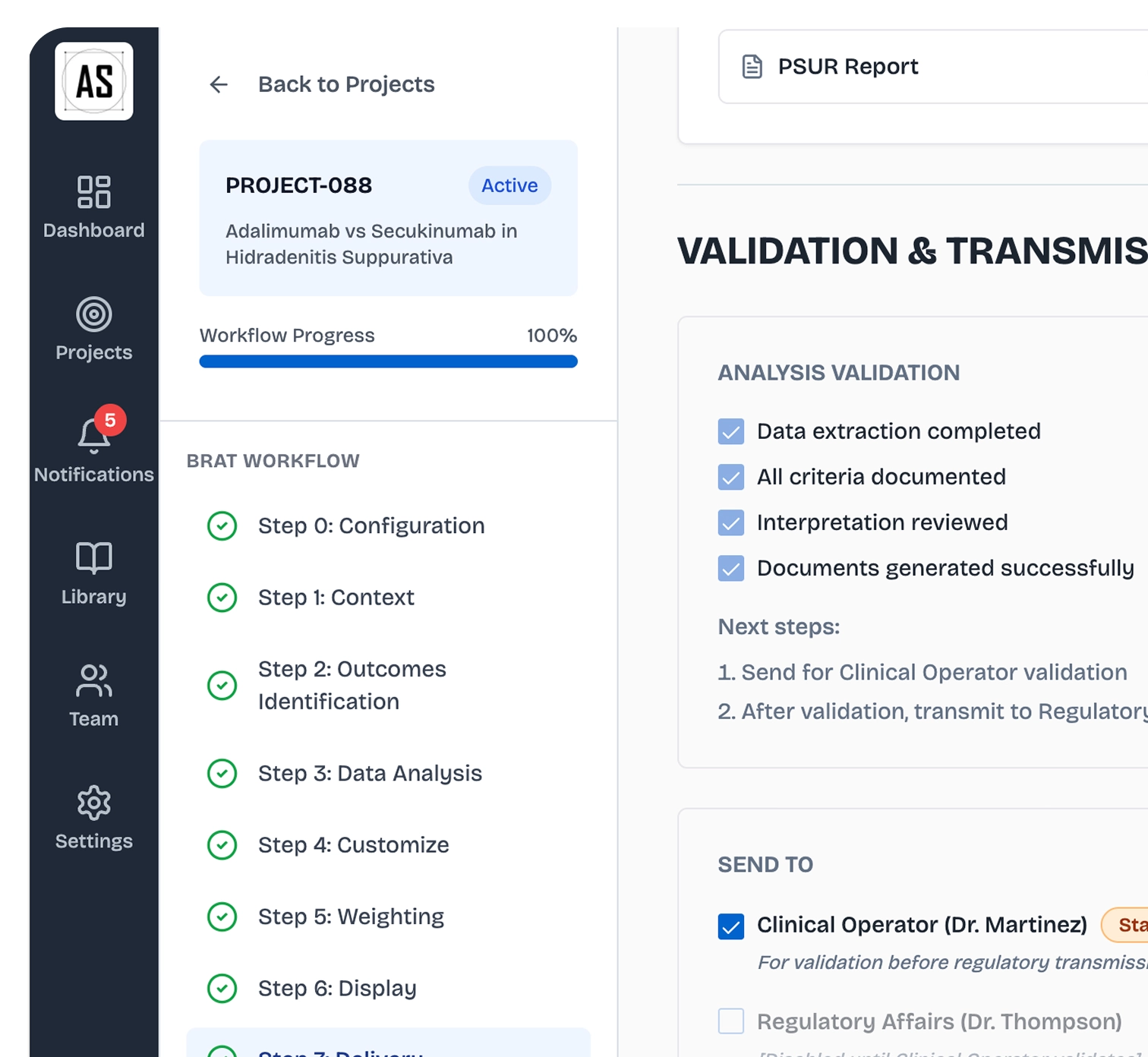
From analysis to submission package
Generate your full regulatory package : PSUR, RMP, CTD 2.5 B-R sections, with complete audit trail. Ready to submit, ready to defend.


Built by scientists.
Trusted by pharmaceutical leaders.
Founded in 2018 by pharmaceutical and medical experts, ArcaScience bridges cutting edge AI with regulatory science. Our platform supports benefit risk decisions across the entire drug lifecycle, from clinical development through post marketing surveillance.
Our Partners
Trusted by leading pharmaceutical companies, research institutions, and regulatory bodies worldwide.




50+ regulatory submissions supported |
12 therapeutic areas
Accelerate Decisions Across the Drug Lifecycle
From early-stage portfolio prioritization to post-marketing safety monitoring, ArcaScience adapts to your specific phase and therapeutic area. Explore how teams like yours leverage our platform at every critical milestone.
The Science Behind the Platform
ArcaScience was built to solve a $13 billion market problem: the lack of comprehensive, real-time benefit-risk intelligence. Our mission is to revolutionize drug evaluation and become the world leader in benefit-risk assessment: replacing legacy service models with transparent, AI-powered decision intelligence.

100B+ Data Points
The world's most comprehensive benefit-risk database (AS Profiling Base 100b®), layering 100% of publicly available scientific literature, RWE, and regulatory submissions.
Peer-Reviewed Methodologies
Published research validating our AI ensemble models for phenotyping, safety signal detection, and comparative effectiveness analysis.
Collaborative Partnerships
Working alongside leading research institutions, regulatory bodies, and pharmaceutical innovators to continuously refine our platform with real-world feedback.
Compliance by design
Pharmaceutical data demands the highest standards. ArcaScience is architected from the ground up to meet global regulatory requirements, ensuring your benefit-risk analyses are audit-ready and legally defensible.




Ready to Transform Your Benefit-Risk Evaluations?
Join 20+ pharmaceutical leaders who've cut evaluation time by 60% and costs by 70%. See how ArcaScience delivers complete benefit-risk intelligence: from clinical framing to automated submissions.










